A Framework for Canine Geroscience
How Do Dogs Age?
A Framework for Canine Geroscience
Introduction
Aging in dogs is not merely the passage of time. It is a progressive biological process characterized by accumulated cellular damage, altered metabolic regulation, immune remodeling, and declining tissue repair capacity.
While companion dogs share environmental exposures with humans, they age more rapidly and exhibit breed-specific lifespan variation. This makes them uniquely valuable both as patients and as translational models for geroscience research.
This article outlines a conceptual framework for understanding canine aging at the biological level.
1. Cellular Senescence
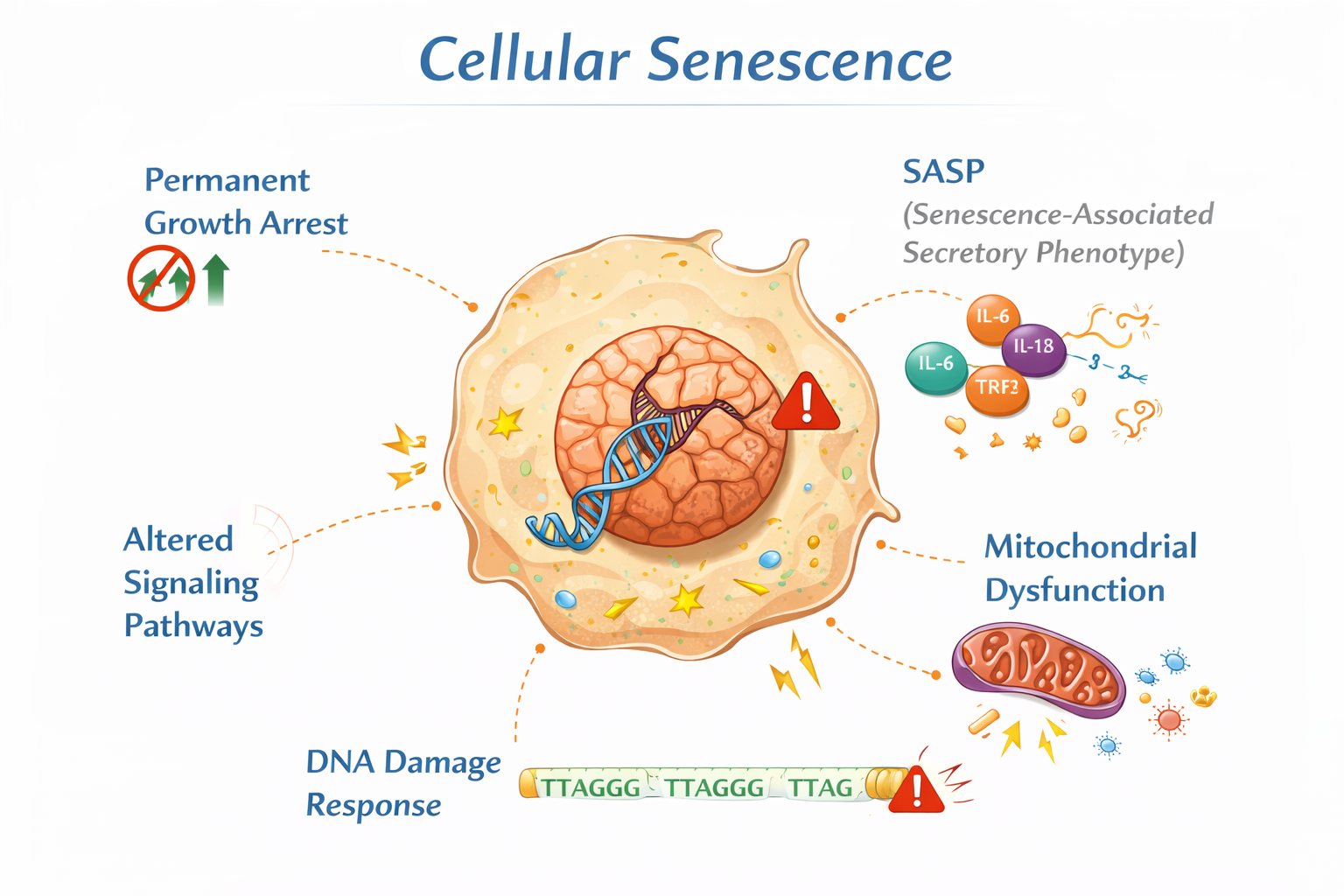
With advancing age, somatic cells accumulate molecular damage from oxidative stress, replication errors, and mitochondrial dysfunction.
Cells may enter a state known as cellular senescence, characterized by:
-
In dogs, increased inflammatory tone and tissue degeneration in older animals suggest similar mechanisms as described in human geroscience.
2. Telomere Dynamics
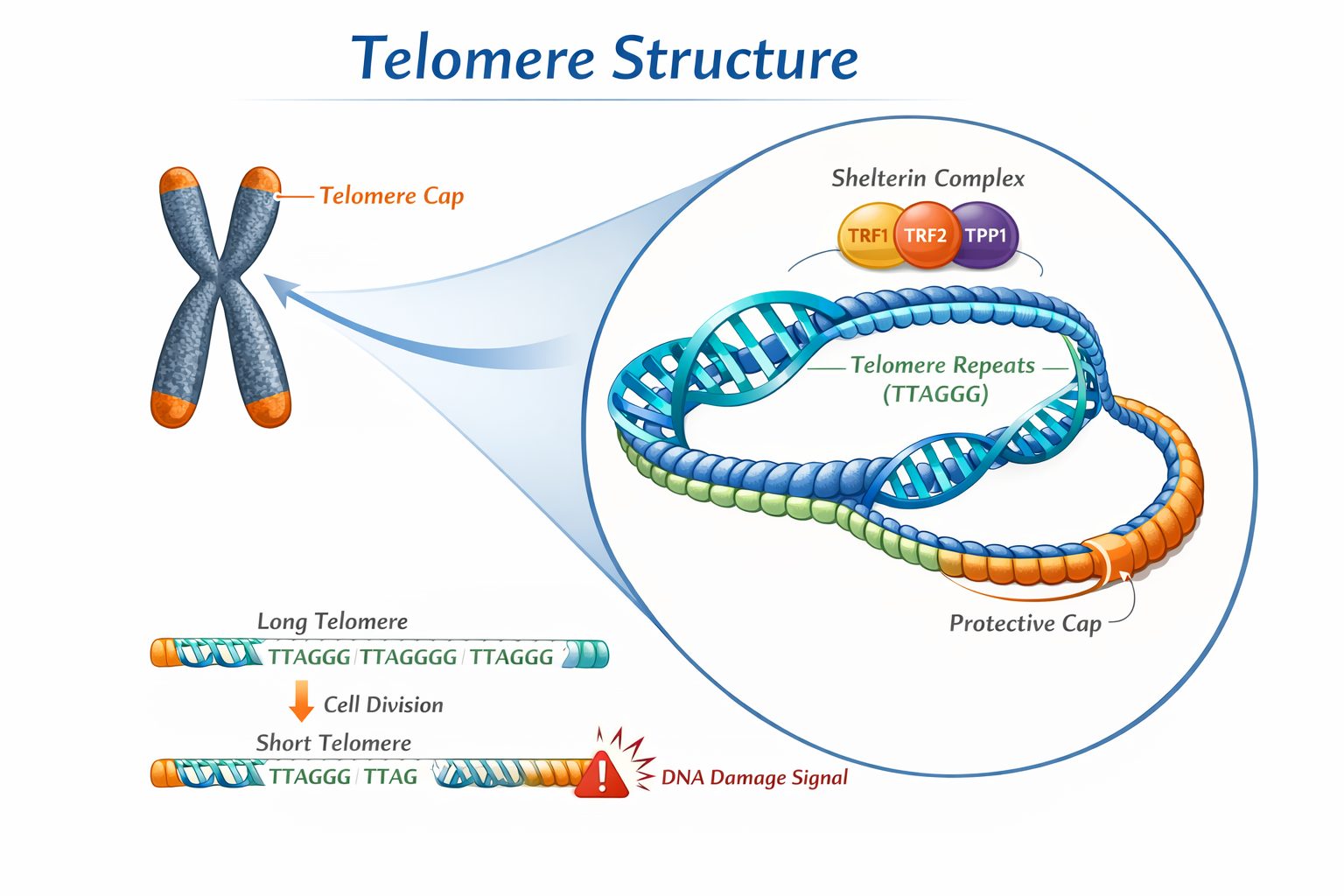
Telomeres are repetitive DNA sequences located at chromosome ends that protect genomic stability.
Over time, telomeres shorten with cell division. Critically short telomeres may:
-
Telomere length measurement (often via qPCR) has been explored as a potential biomarker of aging in dogs, though methodological variability remains a challenge.
3. Epigenetic Alterations
Beyond DNA sequence, aging also involves changes in gene regulation.
DNA methylation clocks have emerged as promising biomarkers of biological age in multiple species. In dogs, emerging research suggests that methylation-based age estimation may provide more precise biological insight than chronological age alone.
Epigenetic drift may reflect cumulative environmental exposure and metabolic stress.
4. Mitochondrial Dysfunction
Mitochondria regulate cellular energy production and redox balance.
Age-associated mitochondrial decline may result in:
-
These changes are linked to sarcopenia, cognitive decline, and immune dysfunction in aging dogs.
5. Inflammaging
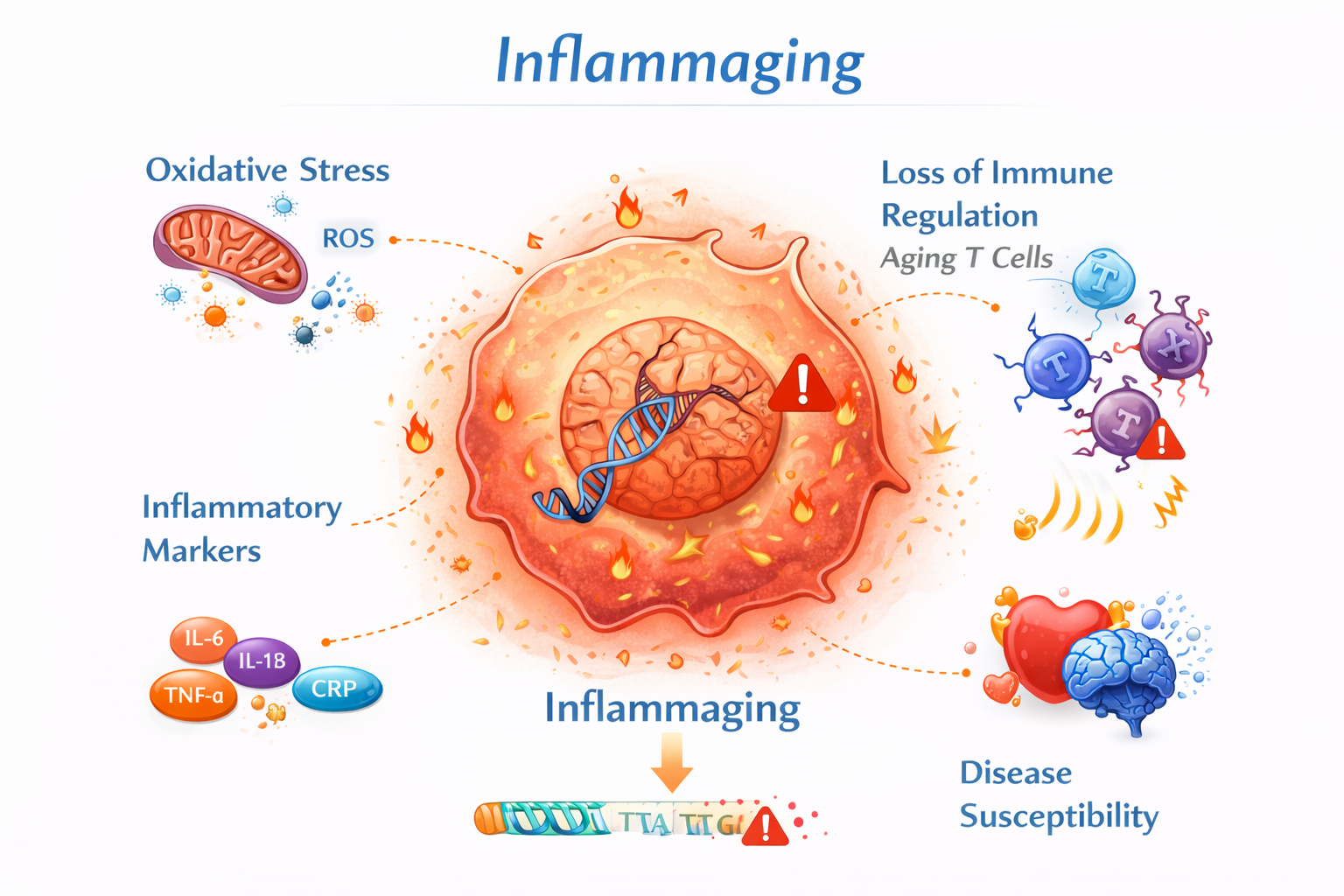
Chronic low-grade inflammation is increasingly recognized as a hallmark of aging.
In senior dogs, elevated inflammatory markers and immune remodeling are associated with:
-
The interaction between immune regulation and metabolic signaling is central to canine geroscience.
6. Musculoskeletal Aging
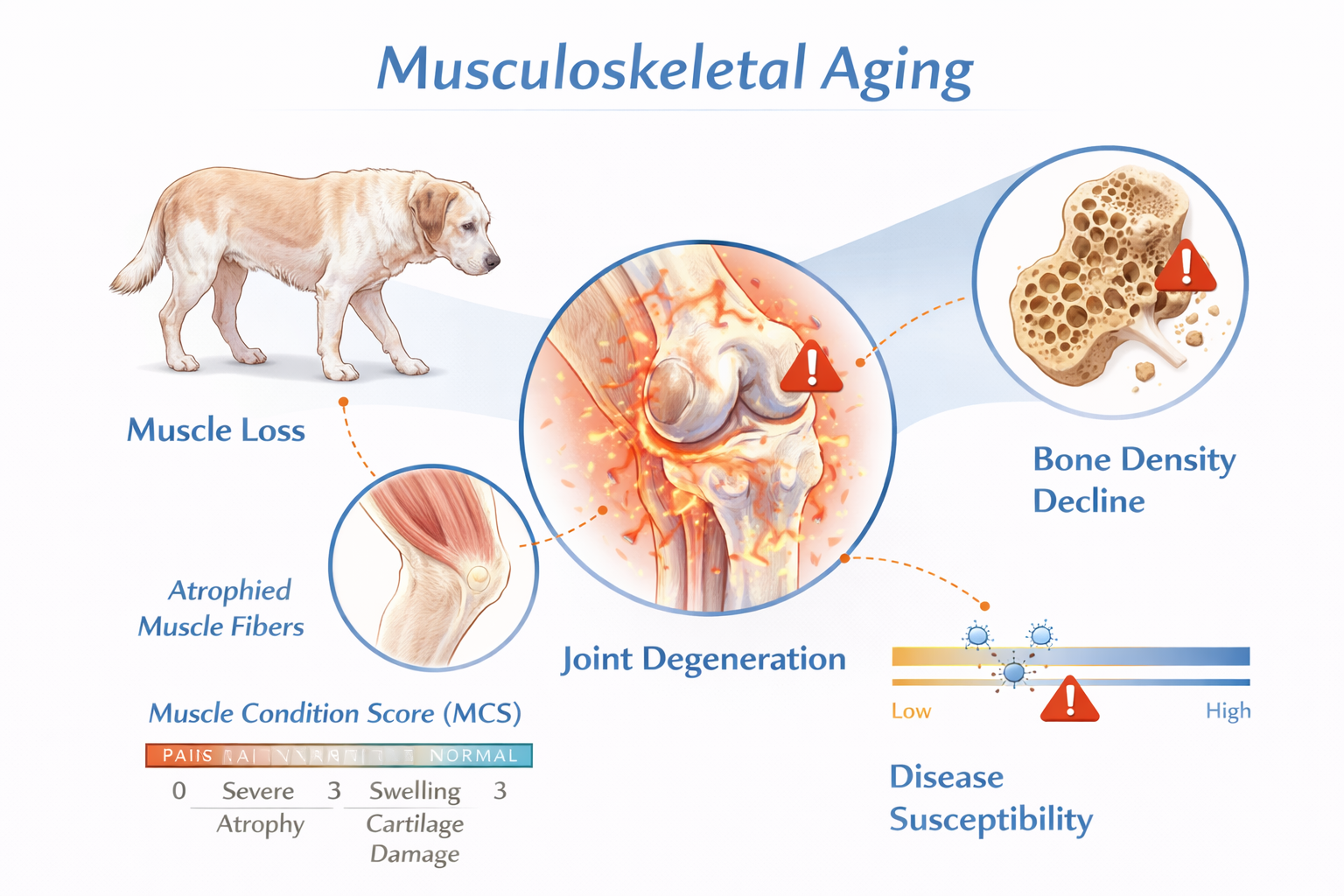
Aging in dogs is clinically visible in:
-
Muscle condition scoring (MCS) and bone mineral density (BMD) assessment provide measurable indicators of structural aging.
7. The Translational Value of Dogs
Unlike laboratory rodents, companion dogs:
-
This makes them a powerful translational bridge between experimental biology and real-world aging interventions.
Programs such as Dog Aging Project have demonstrated the feasibility of longitudinal canine aging research.
Toward a Structured Canine Geroscience Framework
A working framework for canine aging research should integrate:
-
Future interventions—whether nutritional, pharmacological, or biological—should be evaluated within this multidimensional framework.
Canine geroscience is not the pursuit of lifespan extension alone, but the preservation of functional healthspan.